
Uppsala Reports editor
@UMCGlobalSafety

The 20-year journey of the PAHO Pharmacovigilance Network: from shared learning to coordinated action.
When the Pan American Network of Drug Regulatory Harmonisation (PANDRH), representing all regulatory authorities of the Americas, convened a working group of pharmacovigilance regulators in 2005, few imagined that this initiative would become one of the longest-standing regional collaborations in the region.
In the early 2000s, pharmacovigilance strategies and activities across the Americas were fragmented: adverse event (AE) reporting was limited, as was the ability to evaluate reports, pharmacovigilance methods lacked standardisation, and there was little to no coordination among regulatory authorities.
This drove the first meeting of the PANDRH working group on pharmacovigilance in Salvador de Bahía, Brazil, in 2006, marking the beginning of a regional initiative that would redefine collaboration in medicines safety across international borders.
In 2012, the working group officially became the Network of Pharmacovigilance Focal Points of the Americas (PAHO), with its own governance structure, an operational manual, and a community of practice hosted on PAHO’s regional platform on access and innovation for health technologies (PRAIS) platform. Since then, cooperation, capacity building, and shared responsibility have been the foundation of the network.
A close connection and collaboration were also established with the WHO Programme for International Drug Monitoring (PIDM) and Uppsala Monitoring Centre (UMC), with representatives of both institutions often participating in the network's virtual and face-to-face meetings (Figure 1).
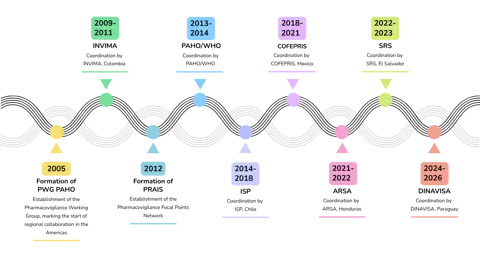
“It was never just about collecting data; it was about building trust among peers, expanding capacities to interpret the data, and taking collective action to protect the population”, says Robin Rojas-Cortés, PAHO’s Technical Officer for regulatory strengthening and safe use of pharmaceuticals.
From its inception, the network was conceived not merely as a discussion forum but as a mechanism encouraging country ownership and structured regional cooperation. Over time, Member States increasingly assumed responsibility for coordinating the network, reflecting growing institutional maturity and shared accountability.
As the network matured, its focus broadened from experience sharing and strengthening spontaneous reporting to helping member states strengthen their capacity to use technical pharmacovigilance tools such as VigiFlow and Vigilyze to conduct signal detection and send data to VigiBase. Regional training initiatives, many conducted with direct participation from UMC experts, improved member states coding practices, MedDRA use, signal detection capacity, and analytical interpretation.
The Data Bridges initiative represents a major milestone in this process. By promoting standardised and automated data transmission, eleven countries have collectively submitted more than 381,000 adverse event reports of AEs and adverse events following immunisation (AEFIs) to VigiBase without having to send or process them individually.
PAHO also developed regional guidance documents and technical tools, established thematic working groups on topics such as signal detection and good pharmacovigilance practices, conducted joint signal discussions drawing on VigiBase data and VigiLyze, and coordinated multi-country surveillance initiatives. Collaboration with national immunisation programmes on the management of AEFIs also became a key focus area, as did strengthening national capacities for high-quality Individual Case Safety Report submission and standardised coding.
As countries in the region worked to reach the WHO Global Benchmarking Tool (GBT) maturity Levels 3 and 4, and some pursued WHO Listed Authority (WLA) status, the network supported them in implementing the WHO GBT pharmacovigilance indicators. PAHO helped foster convergence in pharmacovigilance inspection systems aligned with Good Pharmacovigilance Practices (GVP), as well as helped countries develop structured signal management processes, while aligning with global benchmarking expectations. The structured exchange of safety assessments, often supported by evidence derived from VigiBase, contributed to building trust and reducing duplication of efforts across the region.
The twentieth anniversary of the Network coincided with the 20th (XX) Pharmacovigilance Meeting of the Americas, held in Asunción, Paraguay, in September 2025 under the coordination of the Paraguay National Directorate of Health Surveillance, known as DINAVISA, and PAHO/WHO.
More than a commemorative event, the meeting symbolised the Network’s transition from strengthening reporting systems to advancing proactive, data-driven safety management with discussions ranging from signal detection and digital interoperability to the integration of pharmacovigilance and immunisation programmes. It also served to consolidate the network’s ongoing working groups and formally launch a new regional group on risk communication.
Twenty years after its creation, the PAHO Pharmacovigilance Network of the Americas has grown from a forum for experience exchange into a structured and globally connected regulatory community. By working together, member states have strengthened their reporting systems, improved their signal detection capacity, advanced regulatory maturity, and enhanced the region’s contribution of data to VigiBase.
As the Pharmacovigilance Network enters its third decade, we shift our focus from capacity building to optimising and integrating our pharmacovigilance processes. We wish to improve data quality and interoperability, expand the use of digital and analytical tools, and strengthen collaborative regulatory approaches. Continued engagement with UMC and the WHO Programme for International Drug Monitoring will remain central to this effort.
This article was written in collaboration with PAHO project members Gabriela Carrasco and Robin Rojas Cortés. If you would like to know more about the project, contact rojasedg@paho.org.
Subscribe for more insights, stories, and developments from the PIDM
SubscribeBrazil’s largest immunobiologicals producer shares lessons from integrating WHODrug Global into its clinical and pharmacovigilance workflows at a national scale.
23 April 2026
A Mexican cardiology hospital became the first to establish a dedicated technovigilance centre in a clinical setting, and its work is already contributing to patient safety.
16 April 2026
Two new films document UMC's collaboration with Brazil's Anvisa, exploring how VigiFlow and WHODrug are strengthening pharmacovigilance across the country.
19 March 2026